What does the eye on the battery mean: black, white, red, green
Content
Car owners are not required to know the intricacies of electrical engineering and master the art of experienced accumulators. However, the condition of the battery under the hood is important enough for the reliable operation of the car, and it is desirable to monitor it without spending a lot of time and money on frequent visits to the master.

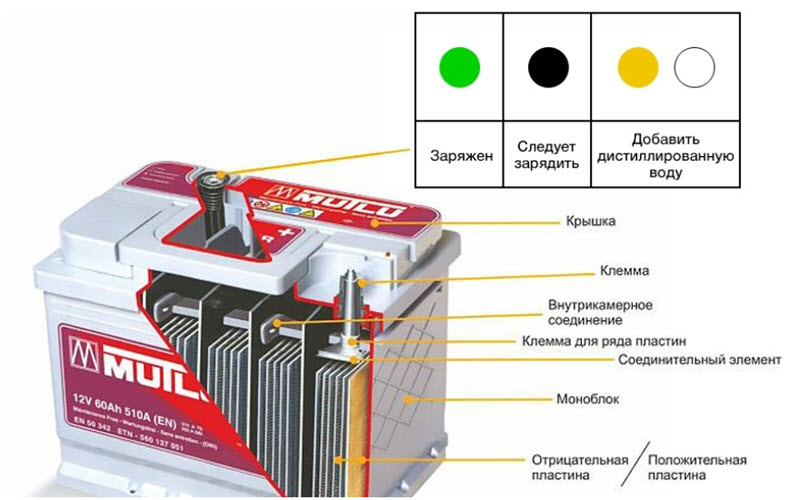
The designers of rechargeable batteries (batteries) tried to get out of the situation by placing a simple color indicator on top of the case, by which one can judge the state of affairs in the current source without delving into the intricacies of the operation of measuring instruments.
Why do you need a peephole in a car battery
The most important criterion for the state of the battery is the presence of a sufficient amount of electrolyte of normal density.
Each element of the battery (bank) works as an electrochemical reversible current generator, accumulating and delivering electrical energy. It is formed as a result of reactions in the active zone of the electrodes impregnated with a solution of sulfuric acid.

A lead-acid battery, when discharged, from an aqueous solution of sulfuric acid forms lead sulfates from oxide and spongy metal at the anode (positive electrode) and cathode, respectively. At the same time, the concentration of the solution drops, and when fully discharged, the electrolyte turns into distilled water.
This should not be allowed, it will be difficult, if not impossible, to completely restore the electrical capacity of the battery after such a deep discharge. They say that the battery will be sulfated - large crystals of lead sulfate are formed, which is an insulator and will not be able to conduct the current necessary for charging reactions to the electrodes.
It is quite possible to miss the moment when the battery is very discharged for various reasons with an inattentive attitude. Therefore, it is recommended to regularly check the state of charge of the battery. Not everyone can do it. But everyone can look at the battery cover and see deviations by the color of the indicator. The idea looks good.
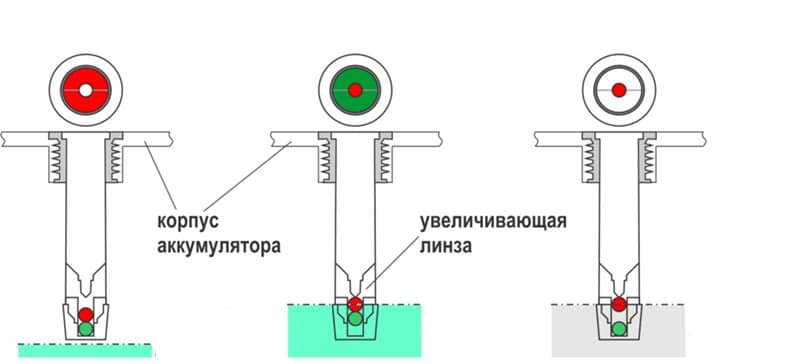
The device is designed as a round hole covered with transparent plastic. It is usually called the eye. It is believed, and this is reflected in the instructions, that if it is green, then everything is fine, the battery is charged. Other colors will indicate certain deviations. In fact, everything is not so simple.
How the battery indicator works
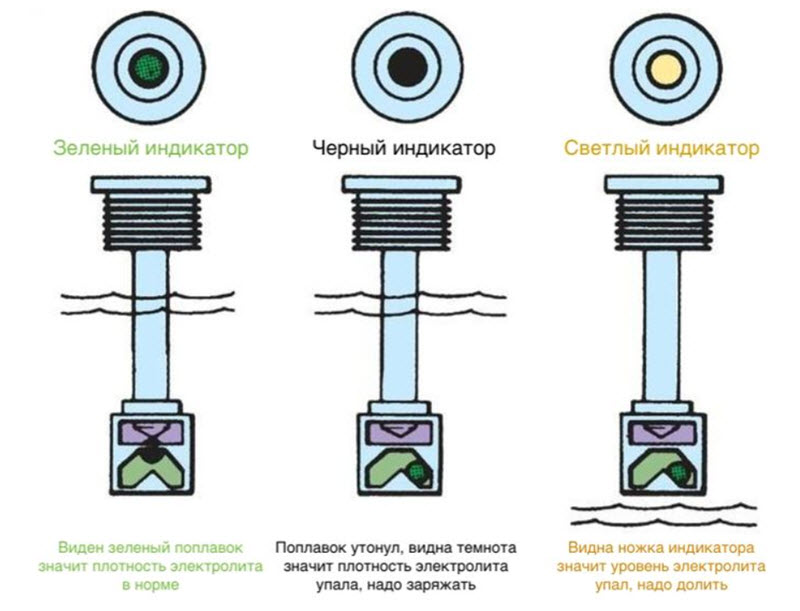
Since each instance of the battery is equipped with an indicator, where it is provided, it was developed according to the principle of maximum simplicity and low cost. According to the mechanism of action, it resembles the simplest hydrometer, where the density of the solution is determined by the last of the floating floats.
Each one has its own calibrated density and will only float in a liquid with a higher density. Heavier ones with the same volume will sink, lighter ones will float.
The built-in indicator uses red and green balls, also having different densities. If the heaviest one has surfaced - green, then the density of the electrolyte is high enough, the battery can be considered charged.
According to the physical principle of its operation, the density of the electrolyte is linearly related to its electromotive force (EMF), that is, the voltage at the terminals of the element at rest without load.
When the green ball does not pop up, the red one is visible in the indicator window. This means that the density is low, the battery needs to be recharged. Other colors, if any, mean that not a single ball floats, they simply have nothing to swim in.
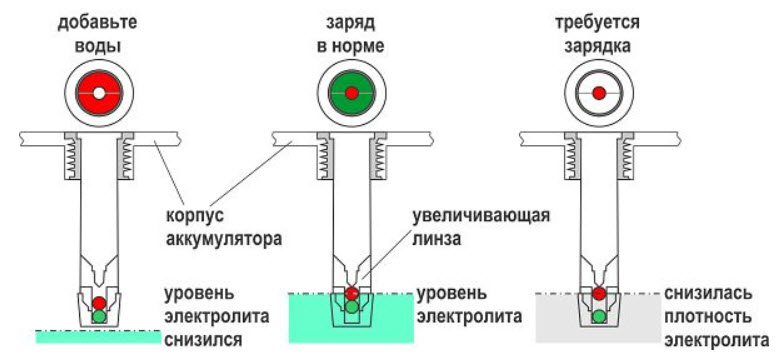
The electrolyte level is low, the battery needs maintenance. Usually this is topping up with distilled water and bringing the density to normal with a charge from an external source.
Errors in the indicator
The difference between an indicator and a measuring device is in large errors, a rough form of readings and the absence of any metrological support. Whether or not to trust such devices is an individual matter.
There are several examples of the inaccurate operation of the indicator, even if it is perfectly functional:
- there is no accounting for the battery temperature, although the ratio between the density of the electrolyte and the degree of charge of the battery largely depends on it;
- the temperature error of the material density of the indicator balls themselves is not certified in any way;
- the battery consists of six independent cells, and the indicator is set to only one of them, the condition of the rest is not even approximately controlled, although one of the most frequent cases of battery failure is precisely the problem with one cell in a serial circuit, and the indicator is placed on one of the middle ones, and most often it is the extreme ones who refuse;
- electrolyte density is far from the only indicator of the state of the battery, including the level of charge;
- in the working area, where all the reactions take place, the density is not measured, and the electrolyte is mixed extremely slowly due to the porous nature of the separators between the electrodes.
If we strictly evaluate the performance of the indicator according to these criteria, then its readings do not carry any useful information at all, since too many reasons lead to their erroneousness.
Color coding
There is no single standard for color coding, more or less essential information is provided by green and red colors.
Black
In many cases, this means a low electrolyte level, the battery must be removed and sent to the table of a battery specialist.
White
Approximately the same as black, much depends on the specific design of the indicator. Do not think, in any case, the battery requires further investigation.
Red
Carries more meaning. Ideally, this color indicates a reduced density of the electrolyte. But in no way should it call for adding acid, first of all, the degree of charge should be assessed and brought to normal.
Green
It means that everything is in order with the battery, the electrolyte is normal, the battery is charged and ready for work. Which is far from a fact for the reasons stated above.

Why is the battery light not on after charging?
In addition to structural simplicity, the device is also not very reliable. Hydrometer balls may not float for various reasons or interfere with one another.
But it is possible that the indicator does indicate the need for battery maintenance. The charge went well, the electrolyte gained a high density, but it is not enough for the indicator to work. This position corresponds to black or white in the eye.
But something else happens - all the banks of the battery received a charge, except for the one where the indicator is installed. Such a run-up of cells in a series connection occurs with long-serving batteries that have not been subjected to cell alignment.
The master should deal with such a battery, perhaps it is still subject to rescue, if it is economically justified. The work of a specialist is quite expensive compared to the prices of budget batteries.
